6 Best LIMS Software in 2026: Top Systems Tested & Compared
Key Takeaways
- LabWare and LabVantage are the strongest choices for large, multi-site enterprise labs in pharma and clinical research that need deep configurability and on-premise deployment.
- Benchling and Scispot.io are best for high-growth biotech and life sciences R&D teams that need a modern API-first LIMS with built-in ELN capabilities.
- The best LIMS software is only as good as the data it relies on. Future-ready labs like Guardant Health, Personalis, and Vanta Diagnostics use Onymos DocKnow alongside their LIMS to capture, validate, and structure data at intake.
The best LIMS software in 2026 connects laboratory information management system workflows to billing, compliance, and downstream operations.
This guide cuts through the noise by comparing the top laboratory information management system software options across clinical, diagnostic, QC, and research use cases so you can make a fast, informed decision.
But remember, your lab’s LIMS is only as powerful as the data feeding it. If paperwork arriving with specimens is incomplete, inconsistent, or manually re-entered, every downstream action carries additional risk. Onymos DocKnow sits upstream of your LIMS to capture, validate, and structure data from the moment it enters your lab.
Compare the Best LIMS Software: At a Glance
| Platform | Best For | Standout Feature |
|---|---|---|
| LabWare LIMS | Large enterprise labs needing heavily customizable, on-prem LIMS | Deep configurability across complex multi-site operations |
| LabVantage Solutions | Enterprise biopharma and clinical research labs | Global multi-site sample management + ELN integration |
| Benchling | Life sciences R&D and biotech labs | Molecular biology tools + ELN tightly integrated with LIMS |
| Thermo Scientific SampleManager | Large pharma/clinical labs needing regulated workflows | Validated compliance modules for FDA 21 CFR Part 11 |
| Scispot | High-growth biotech and genomics labs | API-first architecture with no-code workflow builder |
| Lockbox LIMS | QC labs and smaller specialty labs | Affordable, fast-deploy LIMS for compliance-focused QC workflows |
If you run a diagnostic or clinical laboratory, the platforms above split into two categories:
- General-purpose LIMS adapted for healthcare
- Intake-specific platforms built for labs
Only one platform below was purpose-built for lab accessioning and RCM enablement from the start.
Onymos DocKnow: The Intelligent Intake Layer Before LIMS (Intake-to-Billing Automation)
Onymos DocKnow is an intelligent document processing software and intake layer purpose-built for clinical and diagnostic laboratories, sitting upstream of any LIMS you choose: capturing, validating, and structuring data before it ever reaches your system of record.
DocKnow handles the entire lab accessioning workflow: capturing test requisition forms (TRFs), validating patient and insurance data, reconciling conflicts before they hit downstream systems, and routing structured data directly to billing platforms, RCM software, LIMS, or analytics tools via API.
Guardant Health, RetinaRisk, and Vapotherm are using Onymos to hyperscale their document operations. For labs scaling from 250K to 350K+ specimens annually, where manual workflows physically cannot keep pace, DocKnow is built for exactly that inflection point.
Why this matters for billing
Claims denied due to incomplete patient data or missing physician identifiers at intake can expire before a team has time to appeal them. Timely filing limits of 90 to 180 days mean errors at the front door directly become revenue loss at the back end. DocKnow is designed to stop that chain before it starts.
Onymos DocKnow Key Features
- Intelligent Intake
DocKnow captures TRFs, insurance cards, and supporting clinical documentation from email, cloud storage, fax, or connected systems, then extracts and validates key data using NLP and machine learning. Conflicting or incomplete fields are flagged before anything moves downstream.
→ Learn more about DocKnow’s lab workflow automation for labs that have to use paper.
- Error Prevention at Intake
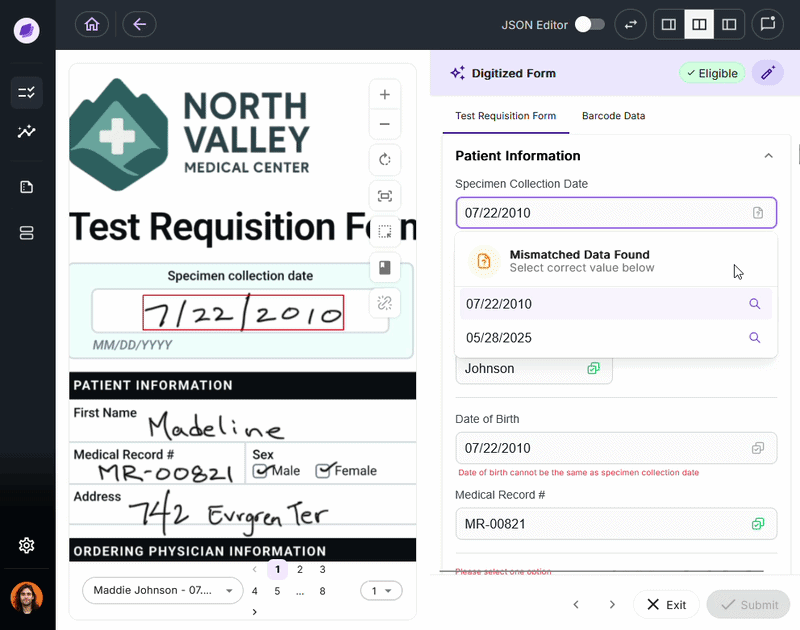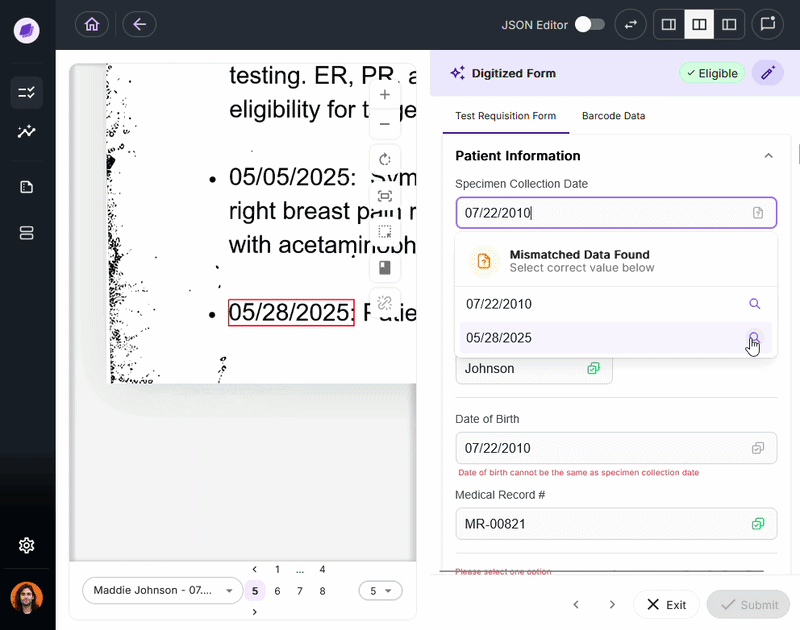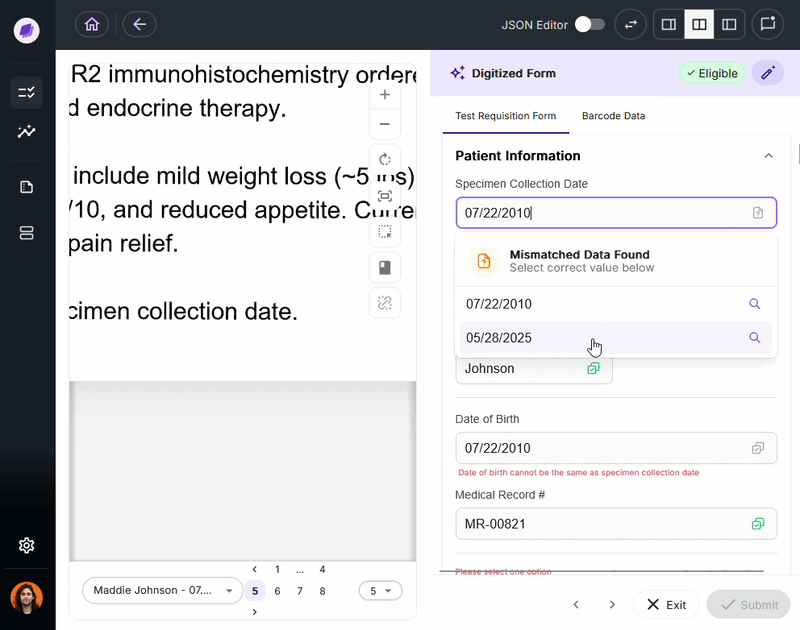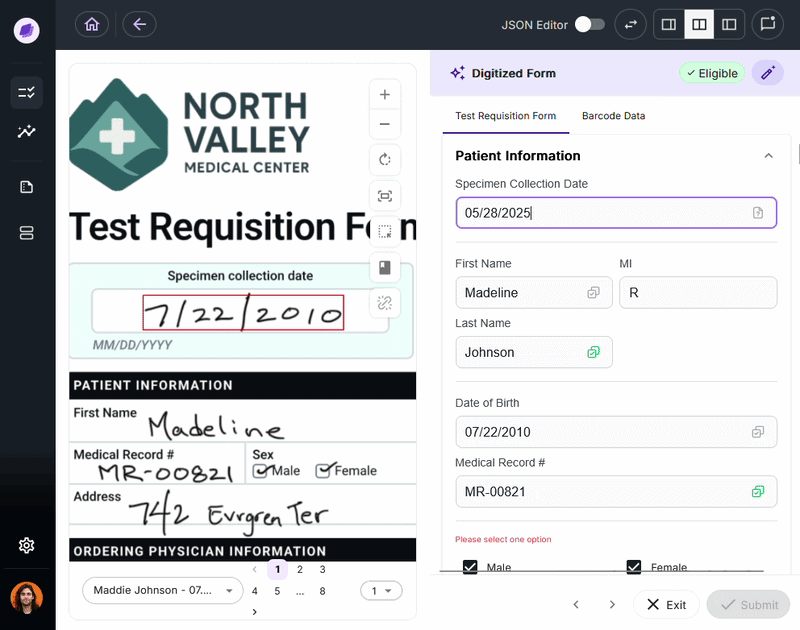
SmartSync is Onymos’s proprietary AI data reconciliation engine, the core of its Nucleus AI system. It cross-references extracted values against connected systems and supporting documents, detecting mismatches.
By integrating into your existing healthcare document management system, SmartSync ensures every document is verified before it reaches downstream processes.
- Billing & Reimbursement Enablement
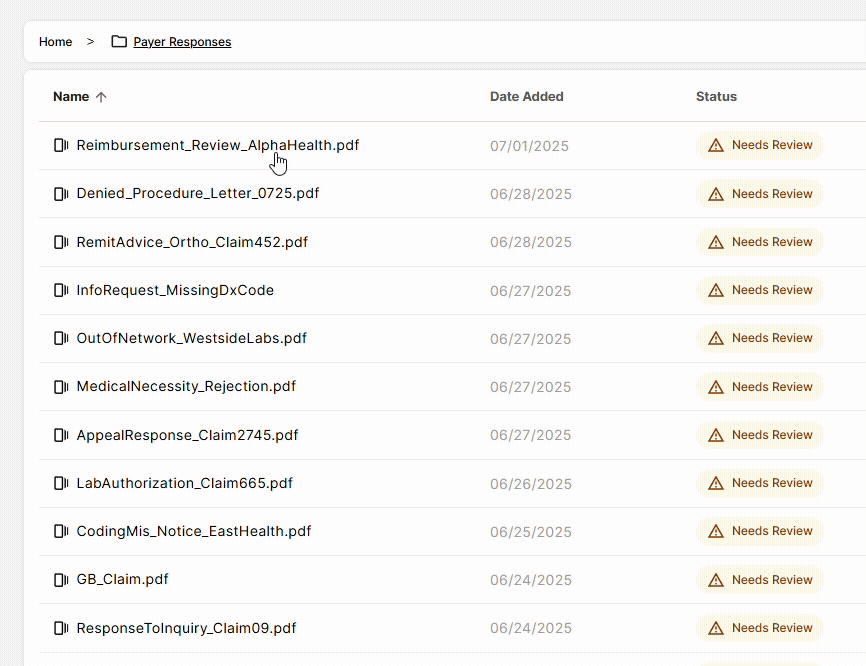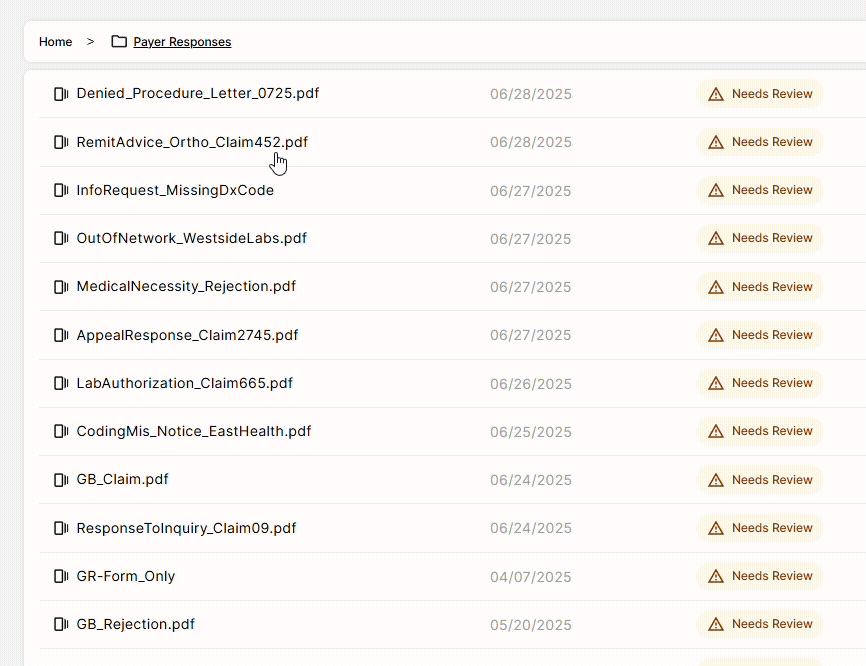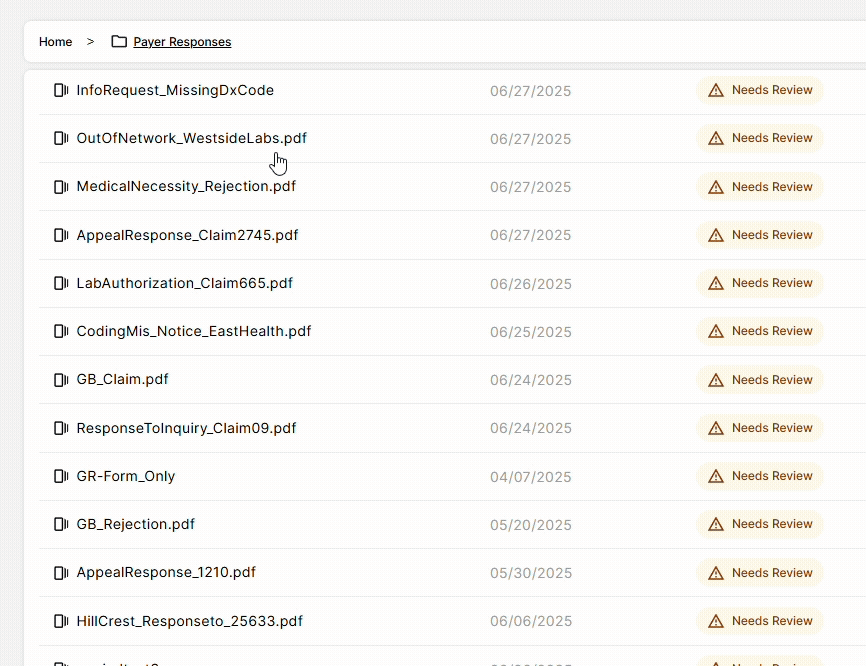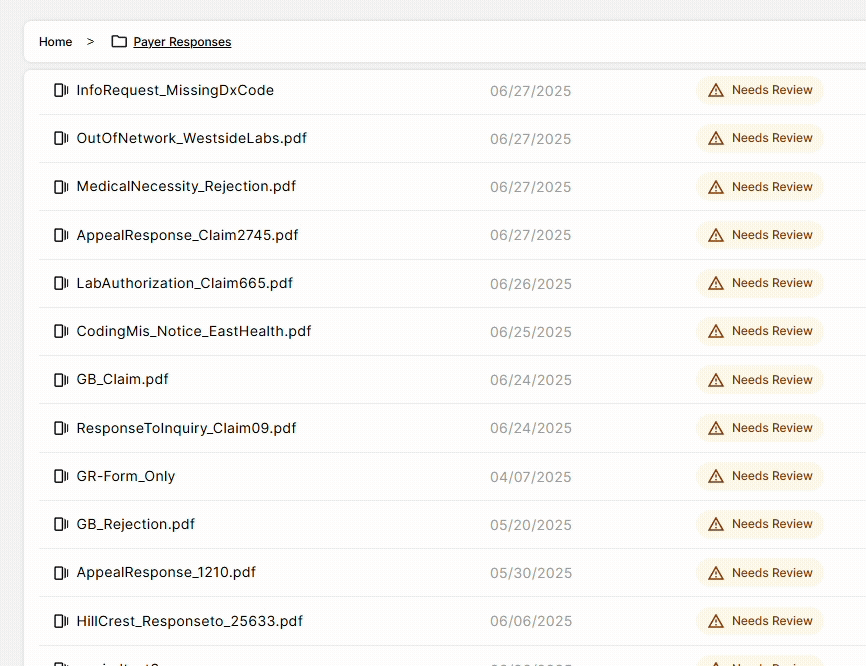
DocKnow also powers the billing and reimbursement workflow, automatically running eligibility checks, processing EOBs and claims, and supporting the generation of appeal letters when denials occur. Every change is logged with a field-level audit trail, keeping your team compliant and audit-ready.
- Security & Compliance
Reports suggest that 80% of stolen patient records originate from third-party vendors. Onymos’s No-Data Architecture addresses this at the infrastructure level: the platform never accesses, captures, or stores customer data, unlike traditional HIPAA-compliant document management software that still relies on data storage models. All patient records, extracted data, and healthcare documents remain exclusively within the customer’s own environment: on-prem or cloud.
→ See Onymos’s full security posture.
Onymos DocKnow Pricing
Onymos DocKnow follows a modular pricing model based on lab volume and workflow complexity. Labs can take advantage of specific features without buying the entire platform.
Where Onymos DocKnow Shines
- Purpose-built for diagnostic and clinical lab intake: Onymos is not adapted from a generic DMS
- SmartSync catches errors at point of intake: This reduces the chances of claim denial
- No-Data Architecture: Drastically cuts the risk of third-party data exposure
- Eligibility checks and appeal letter generation: These functions are built into the billing module
- SOC 2 + HIPAA certified: Compliance is built into the platform to support CLIA/CAP
Where Onymos DocKnow Falls Short
- Primarily focused on labs: Not suitable for general hospital administration
- Not a traditional LIMS: It’s focused on intake and the RCM layer
Customer Reviews
An Onymos user praises, “Onymos is a great partner and enabled us to quickly get our Proof of Concept completed. They were very responsive and collaborative, and we had a successful Proof Of Concept deployment.”
A small business says, “Time to market was extremely important to us. Onymos delivered, allowing us to leapfrog the competition and be the first organization in our medical space with a mobile app.”
→ Read more: Onymos’s customer success stories.
Who Onymos DocKnow is Best For
- Diagnostic labs: Processing high volumes of TRFs insurance cards, and clinical documents at scale
- Clinical labs losing revenue: Due to claim denials caused by incomplete intake documentation
- Lab directors and RCM teams: Anyone responsible for clean data flows from specimen intake through reimbursement
- Labs on growth trajectories: Moving from 250K to 350K+ specimens annually, who can no longer scale manual workflows
Stop letting intake errors become denied claims.
See Onymos DocKnow in action → contact us!
1. LabWare LIMS: Best for Large Enterprise Labs
LabWare Holdings is one of the oldest and most established names in the LIMS space. It’s a highly configurable, enterprise-grade laboratory information management system used by large pharmaceutical, clinical, and industrial labs.
Key Features
- Workflow Depth: LabWare allows labs to configure virtually every workflow, form, and process without writing code.
- Validated Environments: Pre-built modules support FDA 21 CFR Part 11, ISO 17025, and GMP/GLP requirements.
- Enterprise Scale: LabWare is built for large organizations running labs across multiple facilities and geographies, with centralized data management and site-level reporting.
Pricing
LabWare offers enterprise licensing at a custom pricing point.
Where LabWare Shines
- Highly configurable: No coding required
- Compliance support: Focused on regulatory compliance for enterprises
- Comes battle-tested: Especially in large pharma and clinical environments
Where LabWare Falls Short
- Heavy implementation timelines: Often 6-18 months
- Not for RCM-specific workflows: It’s not built for lab accessioning
- Overkill: Especially for mid-size or fast-moving labs
Customer Reviews
One user praises, “The customization that is built into labware platform is amazing. The system is robust and versatile.”
Matthew J. warns, “Since LabWare LIMS can be incredibly customized and specifically designed, it can be challenging when it comes time to upgrade to a new version.”
Who LabWare Is Best For
- Large pharmaceutical and clinical research organizations in regulated environments
- Multi-site enterprise labs in North America, South America, and the Middle East requiring on-prem deployment
2. LabVantage Solutions: Best for Biopharma and Clinical Research
LabVantage Solutions delivers an enterprise laboratory information management system built for biopharma, clinical diagnostics, and manufacturing QC labs. It’s particularly strong in multi-site sample management, biobanking, and ELN integration.
Key Features
- Biobanking and Sample Management: LabVantage handles complex sample hierarchies, chain of custody, and long-term biobanking workflows that most general LIMS platforms can’t manage out of the box.
- Research Workflows: Tight integration with Electronic Lab Notebook (ELN) modules makes LabVantage a strong fit for research-heavy environments where experiment data and sample data need to coexist.
- Global Compliance Support: Supports FDA, EMA, and ISO regulatory requirements across global deployments.
Pricing
LabVantage follows a custom pricing model.
Where LabVantage Shines
- Strong bio banking management: As well as hierarchy management features
- Good ELN + LIMS integration: Offering versatility
- Solid compliance coverage: Offered on a global scale
Where LabVantage Falls Short
- Complex implementation: Not plug-and-play
- Not designed for lab intake: No RCM automation features
- Complex pricing and deployment timelines: Not suited for mid-market labs
See how lab management software supports enterprise R&D scale challenges
Customer Reviews
A user says, “Allows for great cross functional communication and lab tracking. Results are easy for groups to see and they’re always accessible. If I need to find a sample owned by more regular users, it is very quick.”
Mohamed S. notes, “Real time integration is not possible.”
Who LabVantage Is Best For
- Biopharma and clinical research labs that manage complex sample hierarchies and need ELN integration
- Enterprise organizations with multi-site, globally distributed laboratory operations
For teams using broader integration stacks, iPaaS tools like MuleSoft competitors such as Workato and Boomi are often compared with API-first architectures like this.
3. Benchling: Best for Life Sciences R&D and Biotech
Benchling has become the modern standard for high-growth biotech and life sciences R&D teams. It’s less a traditional LIMS and more a unified platform combining molecular biology tools, an ELN, and LIMS capabilities.
Key Features
- R&D-Native: Benchling includes sequence editors, plasmid maps, and molecule registration tools built natively into the platform.
- Unified Research Platform: Research data and sample tracking coexist in a single system, reducing the need for separate ELN and LIMS subscriptions.
- API-First Architecture: Robust API connectivity enables Benchling to plug into existing data infrastructure, analytics platforms, and lab automation systems.
Pricing
Benchling offers academic and small teams a free tier. Growth and Enterprise firms need to follow a custom model.
Where Benchling Shines
- Best-in-class for life sciences: Especially in the R&D and biotech workflows space
- Free tier available: Rare in this market
- Modern UI: Offering fast onboarding
Where Benchling Falls Short
- Not built for diagnostic lab accessioning: Or clinical workflows
- Unsuitable for high-volume: Firms operating in busy clinical environments won’t find it a good fit
- Enterprise pricing is costly: Scales up quickly
Customer Reviews
Andrew L. shares, “I love the integration of an electronic lab notebook with a suite of tools for molecular cloning. As a molecular biologist, I use Benchling constantly and it has made me a better scientist.”
One user notes, “Sometimes it can be a bit slow, specially when trying to upload gel images onto the notebook entry. The sequence data analysis feature is a bit basic, and doesn’t let you link specific sequences to particular entities in your notebook entry.”
Who Benchling Is Best For
- Biotech and life sciences R&D teams needing modern molecular biology tools alongside LIMS capabilities
- Academic and startup labs that want a free or low-cost entry point with strong API integration
4. Thermo Scientific SampleManager: Best for Regulated Pharma Labs
Thermo Scientific SampleManager (part of Thermo Fisher Scientific Inc.) is a mature, validated LIMS platform built for pharmaceutical and clinical labs operating in highly regulated environments.
Key Features
- Regulatory-Grade: SampleManager offers validated software packages for FDA 21 CFR Part 11, Annex 11, and GMP environments. This is a critical differentiator for pharma labs that cannot afford to fail an audit.
- Lab Efficiency: Built-in QC workflow automation capabilities reduce manual review steps and improve turnaround times for regulated testing processes.
- Automation Capabilities: SampleManager supports broad instrument integration, enabling automated data capture from lab instruments directly into the LIMS.
Pricing
Thermo Scientific offers custom pricing for all enterprise tiers.
Where Thermo Scientific Shines
- Best-in class: For validated, regulated pharma environments
- Strong automation: In the QC workflow processes
- Integrations: Broad instrument integration and automation capabilities
Where Thermo Scientific Falls Short
- Customization burden: May take substantial effort
- Heavy efforts required: In implementation and validation
- Overkill: For labs outside regulated pharma or clinical research
Customer Reviews
Pratibha S. shares, “I’m currently working in Food Industry as Analyst and often used Thermoscientic LIMS. It’s a one stop solution for the laboratory to manage the data SOP, Records , formats and complies with Quality Assurance.”
Amit K. noted that lab processing functions are disturbed if any server disruptions occur.
Who Thermo Scientific Is Best For
- Large pharmaceutical and clinical labs in regulated environments requiring validated compliance modules
- Organizations prioritizing instrument integration and automation capabilities at scale
5. Scispot: Best for High-Growth Biotech and Genomics Labs
Scispot.io is an emerging, API-first laboratory information management system built for fast-moving biotech, genomics, and precision medicine labs. Scispot is designed to deploy quickly and integrate with modern data stacks via its no-code workflow builder and robust API.
Key Features
- Rapid Deployment: Labs can build and modify workflows without engineering resources.
- Modern Integration: Scispot’s API enables deep integration with sequencing platforms, analytics tools, and cloud infrastructure.
- Lab Operations: Built-in inventory management tracks reagents, consumables, and equipment across lab operations, reducing manual stockroom management.
Pricing
Scispot also offers custom pricing for all enterprise tiers.
Where Scispot Shines
- Fast deployment: With a no-code workflow builder
- Modern data integration: Made possible through strong API-first architecture
- Great for genomics: Also a good fit for precision medicine labs
Where Scispot Falls Short
- Newer platform: Less proven at large enterprise scale
- Not built for clinical lab accessioning: Better aligned to research
- Limited regulatory validation modules: Compared to Thermo Scientific SampleManager
Customer Reviews
Shiva Raju G. praises, “Scispot team has resident scientists and computational biologist who helps us write custom scripts, configure custom schema for databases, and give suggestions on workflow automation and data strategy.”
Soumith P. notes, “I would like to see more scientific out of the box analytics.”
Who Scispot Is Best For
- High-growth biotech and genomics labs that need a fast-deploying, API-first LIMS
- Precision medicine and sequencing labs with modern data infrastructure
6. Lockbox LIMS: Best for QC Labs and Specialty Labs
Lockbox LIMS is a focused, compliance-oriented laboratory information management system designed for QC labs, environmental testing labs, and specialty labs that need a straightforward, affordable LIMS without the enterprise overhead.
Key Features
- QC Focus: Lockbox LIMS is built around QC lab workflows, making it a natural fit for food, environmental, and manufacturing QC environments.
- Low Implementation Burden: Lockbox is designed to deploy quickly which is important for smaller labs that can’t absorb lengthy IT projects when adopting new lab workflow software.
- Core LIMS: Core sample tracking and inventory management capabilities cover the fundamentals most QC labs need without the premium tiers.
Pricing
LockBox offers modular pricing. The provider notes the average LIMS price for most subscriptions is ~$225/user/month,
The first module of the LIMS is priced at $175/user/month, and each additional module is $25/user/month.
Where Lockbox Shines
- Accessible public pricing: Rare in the LIMS market
- Fast deployment: Especially for QC labs
- Strong COA and specification management: Great for QC focused processes
Where Lockbox Falls Short
- Limited advanced features: Especially compared to enterprise platforms
- Limited integrations: Smaller ecosystem compared to Benchling or Scispot
- Salesforce dependency: Risk of vendor lock-in
Customer Reviews
Lindsay K. shares, “Lockbox worked closely with our team to understand our processes and procedures, then created flows for our very unique and complex testing environment.”
Nicolai L. warns, “The initial setup of Lockbox LIMS was a lot of work.”
Who Lockbox Is Best For
- QC labs and environmental testing labs that need a fast-deploy, lab compliance ready LIMS
- Smaller specialty labs that want public pricing and a low implementation burden
Why Even the Best LIMS Software Needs Onymos DocKnow
Most LIMS platforms handle the science side well. The problem isn’t what happens inside the system; it’s what arrives at the door.
Incomplete TRFs, mismatched patient records, and manually re-entered data don’t get fixed by a better LIMS. They get fixed upstream.
Here’s how DocKnow addresses each layer of that problem.
1. DocKnow Understand Your Documents, Not Just Your Data Fields
A generic intake process treats every document the same. DocKnow doesn’t. It’s built to understand the specific document types that drive lab operations:
- Test requisition forms
- Insurance cards
- Chain-of-custody records
- Clinical documentation
It extracts, validates, and structures that data in context, so what reaches your LIMS is already clean, complete, and system-ready.
Need intake-to-billing automation built for labs?.
See how Onymos DocKnow connects your intake workflow to reimbursement → contact us!
2. No-Data Architecture Means Your Patient Data Never Leaves Your Environment
Security in healthcare isn’t just about encryption. HIPAA compliance is the baseline, but the deeper question is: does your vendor’s platform ever touch your data at all?
DocKnow is built on Onymos’s No-Data Architecture, which means the platform is deployed within your own infrastructure. Patient records, extracted data, and clinical documents never pass through Onymos servers.
Your data stays yours, whether you’re on-prem or in the cloud.
3. Built for the People Running the Lab, Not Just the People Buying the Software
Lab technicians, specimen handlers, and billing teams are not IT professionals. A system that requires significant training or IT support to run daily workflows will create more problems than it solves.
DocKnow’s interface is designed for the people actually doing intake with:
- Visual mismatch alerts
- Simple correction flows
- Configurable business rules that don’t require engineering support to maintain
If it creates friction for your frontline staff, it won’t get used. DocKnow is built so it will.
4. Structured Output That Plugs Into Whatever Stack You’re Running
DocKnow doesn’t replace your existing systems but it can feed into them. Validated, structured data is delivered via API in standard formats including FHIR and JSON, routing directly to your LIMS, EHR, billing platform, or analytics stack.
Say goodbye to manual re-entry and translation layers. You get clean data, where it needs to go, from the moment intake begins.
The Best LIMS Decision You Can Make Is Fixing What Comes Before It
You’ve compared the options. You know which LIMS fits your lab’s size, compliance requirements, and workflow. But whichever platform you choose, it will only perform as well as the data you feed it.
DocKnow is the intake layer that sits upstream of it all. It works alongside LabWare, LabVantage, Benchling, or any other system on this list, capturing, validating, and structuring your documents before they touch your LIMS, billing platform, or analytics stack.
Whatever LIMS you run, run cleaner data through it first.
See how DocKnow works alongside your LIMS.
For more on how lab intake connects to billing outcomes, explore our guide.