HITRUST vs HIPAA: Key Differences Explained (2026 Guide)

Key Takeaways
- HIPAA is a federal legal requirement for covered entities and business associates, while HITRUST is a certifiable security framework used to demonstrate structured compliance and security rigor.
- HITRUST does not replace HIPAA. Even with HITRUST CSF certification, organizations are still legally required to comply with HIPAA, and certification alone does not provide a legal safe harbor.
- Most healthcare organizations and labs must comply with HIPAA, but HITRUST is typically optional unless required by enterprise customers such as large health systems, payers, or PBMs—where HITRUST i1 or r2 is often contractually mandated in 2026.
- The key distinction is that HIPAA defines what must be protected, while HITRUST provides a structured, auditable way to prove how those protections are implemented across 60+ aligned security and privacy frameworks.
Most healthcare organizations understand HIPAA. They know it governs protected health information, they know violations are expensive, and they know their business associates are in scope.
What creates confusion is HITRUST: a framework that references HIPAA extensively, overlaps with it substantially, and yet is a completely separate thing.
The short answer:
- HIPAA is the law.
- HITRUST is a certification framework that helps organizations demonstrate HIPAA compliance in a structured, independently verifiable way.
- HITRUST CSF does not replace HIPAA. HIPAA compliance does not confer HITRUST certification.
This guide breaks down what each one actually is, how they interact, where they diverge in ways that matter to real compliance decisions, and whether your organization needs one, the other, or both.
What Is HIPAA?
HIPAA (the Health Insurance Portability and Accountability Act) is a US federal law enacted in 1996 that establishes national standards for protecting the privacy and security of certain health information.
It was originally designed to make health insurance portable between jobs, but the provisions that dominate healthcare compliance today are in Title II: the Privacy Rule, the Security Rule, and the Breach Notification Rule.
HIPAA applies to two categories of organizations:
- Covered entities (such as health plans, healthcare clearinghouses, and healthcare providers) that transmit health information electronically are directly subject to HIPAA
- Business associates (which include any vendor or contractor that creates, receives, maintains, or transmits protected health information (PHI) on behalf of a covered entity) are subject to HIPAA through their Business Associate Agreements (BAAs) and, since the HITECH Act, directly under the law itself
The distinction between these two is where most practical compliance questions arise. A diagnostic laboratory is typically a covered entity.
A software vendor processing lab documents on behalf of that lab is a business associate. Both are technically in scope and bear direct regulatory risk.
HIPAA compliance requires the following at a functional level:
- Documented policies and procedures
- A risk analysis
- Workforce training
- Access controls
- Audit logs with encryption where appropriate
- Breach notification processes
- Executed BAAs with all relevant vendors
While HIPAA does not specify exactly how to implement most of these controls, it sets the standard but leaves implementation to the organization.
A HIPAA-compliant document management system is one of the most operationally significant pieces of the HIPAA security posture for labs and healthcare organizations that handle high volumes of clinical documents.
Understanding healthcare data security more broadly also sets the context for why both HIPAA and HITRUST exist.
What Is HITRUST?
The most important thing to know from the outset is that HITRUST is not a law, a government agency, or a regulation. It is a private organization (the HITRUST Alliance) that created and maintains the HITRUST Common Security Framework (CSF), a certifiable security and compliance framework that brings together over 60 regulatory standards and frameworks into a single control set.
It was created in 2007 specifically to address a problem that healthcare organizations kept running into: every enterprise customer, payer, and partner had different security questionnaires, different audit requirements, and different evidence expectations. HITRUST covers HIPAA, NIST 800-53, ISO 27001, PCI DSS, GDPR, and dozens of other frameworks simultaneously to solve that problem.
When an organization achieves HITRUST certification, it’s been validated by a HITRUST-authorized external assessor and demonstrated that its controls meet the HITRUST CSF requirements at the relevant maturity level.
That certification is then recognized by enterprise healthcare customers as evidence of security rigor, giving them more confidence and reducing the need for individual security questionnaires in vendor procurement.
HITRUST is not a single binary pass/fail certification. There are three assessment tiers to know:
| Assessment | Controls | Validity | Best For |
|---|---|---|---|
| e1 | 44 foundational controls | 1 year | Low-risk organizations, first-time HITRUST credential |
| i1 | 182 implemented controls | 1 year | Mid-risk organizations, enterprise healthcare vendor contracts |
| r2 | 250 to 1,000+ risk-based controls | 2 years | High-assurance, complex environments, payer/PBM contracts |
If a healthcare enterprise customer contract says “HITRUST certification required” without specifying a level, procurement almost always means i1 or above. Certifying at e1 when the customer requires i1 means redoing the work within months. Clarify the required tier before you start.
For labs at risk of third-party supply chain exposure, HITRUST certification provides documented assurance that a vendor’s controls have been independently tested.
The risks of assuming data protection from AI tools and SaaS vendors make this kind of independent assurance increasingly important.
How HITRUST and HIPAA Work Together
HITRUST does not replace HIPAA. The relationship between them is additive so you should never perceive one as a substitute for the other.
HIPAA defines the legal requirements for PHI protection. The HITRUST CSF provides a structured, independently auditable set of controls that, when implemented, demonstrate compliance with HIPAA along with NIST, ISO, and dozens of other frameworks.
Achieving r2 HITRUST certification generates a detailed map of how an organization’s controls correspond to HIPAA Security Rule requirements, which is exactly what the HITRUST Insights Report provides.
In practical terms:
- An organization can be HIPAA-compliant without being HITRUST certified: this is the default state for most covered entities
- An organization can also be HITRUST certified while still having HIPAA compliance gaps: if their HITRUST scope excluded systems or processes that are in scope for HIPAA
Neither automatically confers the other. What HITRUST adds on top of HIPAA compliance is verifiability. For enterprise procurement teams evaluating hundreds of vendors, this matters enormously.
The key practical points to keep in mind:
- Understanding where your data actually lives is foundational to both HIPAA compliance and HITRUST scoping
- The benefits of No-Data Architecture are relevant specifically here, keeping data inside the customer’s own environment reduces both HIPAA Business Associate risk and HITRUST assessment scope
- Fake data ownership versus real data ownership matters to both frameworks
- Why data portability matters is increasingly a factor in how organizations structure vendor relationships to minimize compliance surface area
Key Differences Between HITRUST and HIPAA
The confusion between HIPAA and HITRUST is understandable. They do overlap heavily in healthcare security contexts, and HITRUST explicitly maps to HIPAA requirements. But the differences between them are consequential for compliance strategy, procurement decisions, and vendor management.
Here is a table first, then the most decision-relevant differences in depth.
| Factor | HIPAA | HITRUST |
|---|---|---|
| Legal authority | Federal law, enforced by OCR/HHS | Private framework, no regulatory authority |
| Who sets the standard | US Congress / HHS rulemaking | HITRUST Alliance |
| Mandatory vs. voluntary | Mandatory for covered entities and business associates | Voluntary but increasingly contractually required |
| How compliance is verified | Self-attestation + OCR audits/investigations | Independent external assessor validation |
| Scope of coverage | PHI privacy, security, breach notification | 60+ frameworks including HIPAA, NIST, ISO, PCI, GDPR |
| Certification exists? | No | Yes — e1, i1, r2 |
| Cost of non-compliance | Civil penalties up to $2.19M/year per tier; criminal penalties up to $250K and 10 years | No regulatory penalty; reputational and contractual consequences |
| Update cadence | HHS rulemaking (infrequent) | HITRUST Alliance updates CSF regularly (v11.7.0 current as of 2026) |
Legal Obligation vs. Market Credential
The most fundamental difference between HIPAA and HITRUST is their legal status.
HIPAA is a federal law. If you are a covered entity or business associate under HIPAA and you fail to comply, the Office for Civil Rights can investigate, levy civil monetary penalties and refer cases for criminal prosecution. This is not optional compliance. There is no self-exclusion.
HITRUST certification, by contrast, is voluntary. No federal agency requires it. No law mandates it. The consequences of not having HITRUST certification are market and contractual. You might lose vendor contracts and cannot pass enterprise procurement requirements. You spend more time and resources answering individualized security questionnaires from each customer.
That said, “voluntary“ understates how functionally mandatory HITRUST has become in certain healthcare contexts. Most enterprise health plan and hospital system vendor contracts now specify HITRUST i1 or r2. Not having HITRUST certification is increasingly disqualifying even though no law requires it.
Prescriptive Law vs. Comprehensive Control Framework
HIPAA establishes what must be protected and what categories of safeguards are required (administrative, physical, technical) but it is deliberately not prescriptive about how to implement them.
The HIPAA Security Rule identifies required and addressable specifications, but the “addressable” designation means an organization can choose an alternative implementation if it is reasonable and appropriate. This creates significant variability in what “HIPAA compliant” actually means across different organizations.
HITRUST CSF is the opposite of flexible. The HITRUST CSF, particularly at the r2 level, specifies controls at a granular implementation level, requires documented evidence of each control’s implementation, and evaluates organizations across five maturity levels: policy, procedure, implemented, measured, and managed.
This difference is why HITRUST certification carries more weight in enterprise procurement than a HIPAA self-attestation. When a health system procurement team asks for proof of security controls, a signed BAA plus a HIPAA self-assessment answers the legal minimum.
The HITRUST CSF vs HIPAA distinction on this dimension is also where HITRUST adds the most value beyond HIPAA: it provides a common, independently verifiable language for communicating security posture to partners, customers, and regulators without requiring every counterparty to conduct their own audit.
Enforcement Mechanism and Penalty Structure
HIPAA violations are enforced by HHS’s Office for Civil Rights through investigations triggered by complaints, breach notifications, and compliance audits. Penalties are tiered based on culpability from situations where the entity did not know (and could not reasonably have known) of the violation to situations involving willful neglect.
HITRUST has no enforcement mechanism in the regulatory sense. The HITRUST Alliance cannot fine organizations, initiate investigations, or impose penalties. What it can do (and does) is revoke certification if an organization fails its interim assessment or annual recertification.
HIPAA non-compliance creates regulatory exposure that can result in significant financial and criminal liability. HITRUST non-compliance creates market exposure. Both are consequential but they operate through entirely different mechanisms.
Do You Need HITRUST If You’re Already HIPAA Compliant?
It depends:
- If your customer base includes large health systems, payers, or PBMs, you likely need HITRUST i1 or r2 in addition to HIPAA compliance.
- If your customer base is primarily smaller providers or organizations that do not contractually require HITRUST, HIPAA compliance may be sufficient for now.
But there are key factors that push the answer in one direction or the other.
You Probably Need HITRUST If…
- Your enterprise customer contracts specify HITRUST certification (most large health plan and hospital system vendor agreements now do)
- Your sales cycle regularly stalls at the security questionnaire stage because you cannot provide a certification
- You are selling into PBM relationships, federal contractor workflows, or large health system integrations where r2 is increasingly mandatory under CAA 2026
- You want to reduce the per-customer security review overhead
The HITRUST certification vs HIPAA calculation is primarily a sales and procurement question: what does your customer base require, and what does it cost you to not have certification (lost contracts, extended sales cycles, bespoke security reviews) versus the cost of achieving it?
HIPAA Compliance May Be Sufficient If…
- Your customers are smaller organizations that accept BAAs and self-attestation
- Your vendor relationships do not contractually require HITRUST
- Your organization handles limited PHI and operates at lower risk profile (the e1 assessment may be more appropriate as a starting credential than jumping directly to i1 or r2)
In 2024, environments maintained by HITRUST-certified companies posted a 99.41% breach-free rate. Whether you weigh this as a genuine security outcome or a selection effect, it is the number enterprise procurement teams are increasingly citing in vendor requirements.
How Onymos Supports HIPAA & HITRUST-Aligned Workflows
Onymos DocKnow is an intelligent document processing platform built for clinical laboratory intake, test requisition processing, and revenue cycle workflows, environments where HIPAA compliance is mandatory and HITRUST-aligned security posture is increasingly demanded by enterprise lab customers and health system partners.
Onymos is not a HIPAA compliance tool and is not a HITRUST assessor or certification platform. It is a document and data processing platform whose architecture is specifically designed to reduce the compliance surface area and documentation overhead that HIPAA and HITRUST-aligned programs require.
Onymos Key Features
The three features that most directly reduce compliance burden for labs operating under HIPAA or pursuing HITRUST certification:
No-Data Architecture: Eliminating Third-Party PHI Scope

Most SaaS document processing vendors store or route customer data through their own infrastructure, making them Business Associates under HIPAA and expanding the HITRUST assessment scope for any customer pursuing certification.
Onymos operates differently. No-Data Architecture means Onymos never accesses, stores, or processes patient data on Onymos-controlled infrastructure. All extracted fields and healthcare documents remain exclusively within the customer’s own environment.
For HIPAA purposes, this reduces third-party data exposure risk and simplifies the BAA relationship. For organizations pursuing HITRUST certification, it means Onymos does not expand the customer’s assessment scope the way a typical SaaS data processor would.
SmartSync: Field-Level Audit Trails at Intake
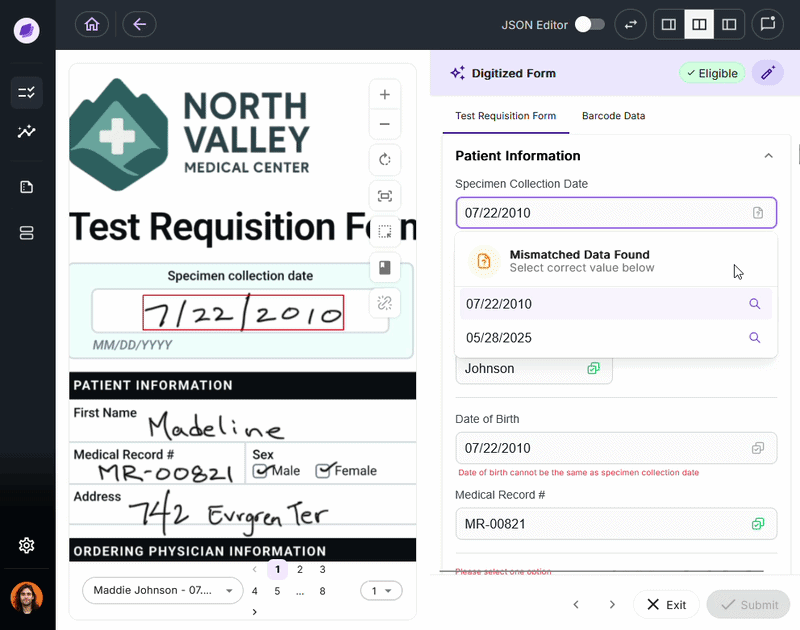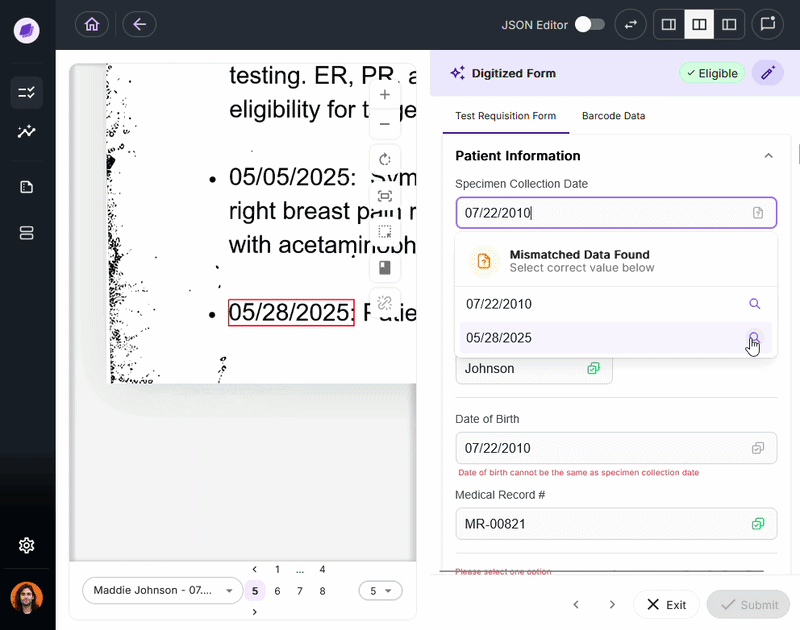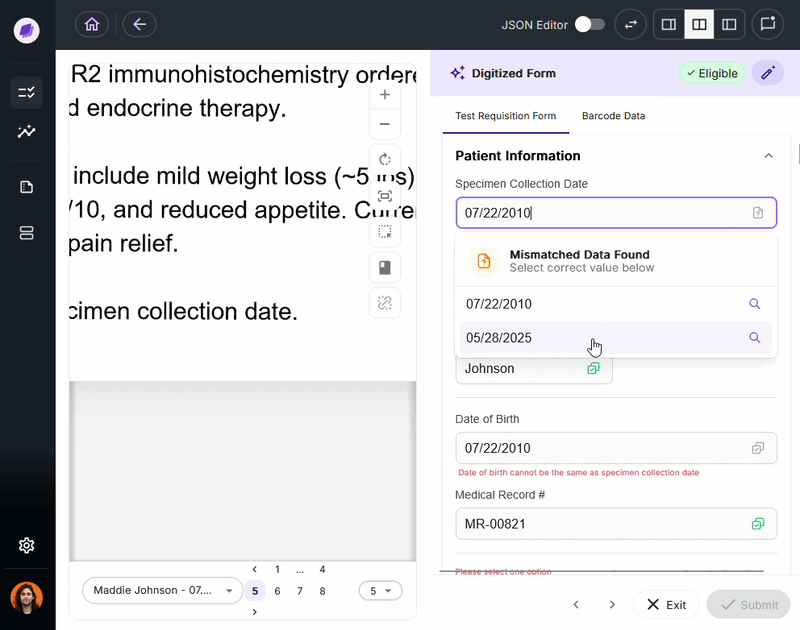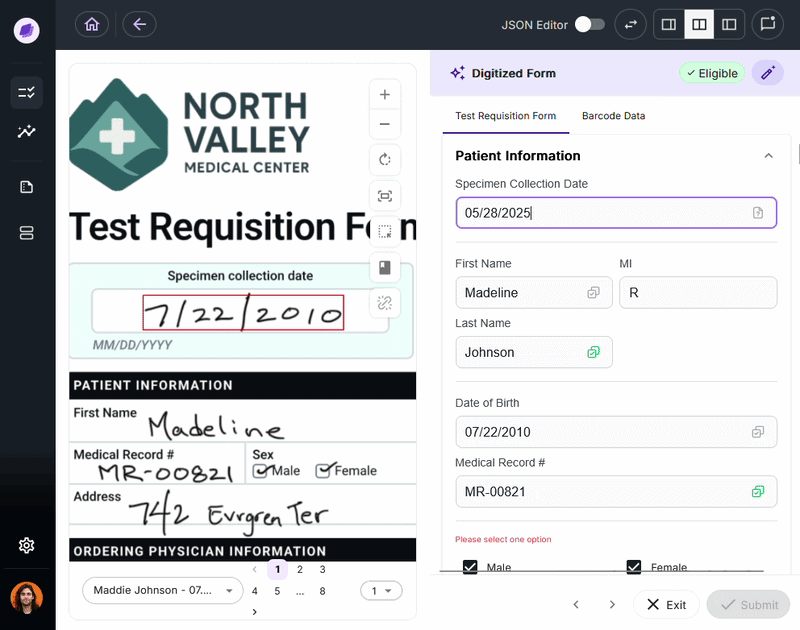
Both HIPAA and HITRUST require organizations to maintain audit trails that log access to and modification of PHI. In practice, maintaining these trails for the messy, unstructured documents that labs process daily is where most manual systems break down.
SmartSync, Onymos’s AI data reconciliation engine, compares extracted field values across connected documents and systems at the point of intake, detecting mismatches and logging every change with timestamps and source attribution functionality increasingly expected from modern lab automation software platforms.
It is built into how the platform processes data, meaning the audit trail that HIPAA and HITRUST-aligned programs require exists in real time, without requiring manual log maintenance by lab staff.
Pre-Built Compliance Artifacts: BAA, SOC 2, Security Documentation
One of the most time-consuming parts of a HITRUST assessment (or any enterprise security questionnaire) is gathering evidence of vendor security controls.
Onymos provides a Business Associate Agreement, SOC 2 Type II report, security documentation, and field-level audit trail capability out of the box, reducing the evidence burden for customers who need to demonstrate their vendor ecosystem’s security posture.
Where Onymos Helps in a HIPAA/HITRUST Program
- Reduces third-party assessment scope: Because patient data never leaves the customer’s environment, Onymos does not expand the customer’s HITRUST assessment surface the way a typical SaaS processor would. Vendors that store PHI add scope to your assessment; Onymos does not.
- Pre-built compliance artifacts ready on request: BAA, SOC 2 Type II report, security documentation, and audit trail capability are available without additional configuration or custom engineering.
- Domain-trained AI for healthcare workflows: Nucleus and SmartSync are built specifically for clinical lab, RCM, and life sciences document workflows, not general-purpose document automation adapted for healthcare. For healthcare document management at the intake layer, that specificity matters for lab compliance as much as it matters for efficiency.
Where Onymos Doesn’t Replace
- Onymos is not a HITRUST CSF assessor: You still need a HITRUST-authorized External Assessor for any validated assessment or certification.
- Onymos is not a compliance platform like Vanta or Drata: It does not manage your policies, run your audits, or track control evidence across your organization. It reduces the surface area those tools have to cover.
- Onymos is healthcare and life sciences focused: Less relevant for general SaaS use cases outside this vertical.
HITRUST vs HIPAA: The Bottom Line
HIPAA is mandatory. If you create, receive, maintain, or transmit protected health information in the US, you comply with HIPAA, and that’s that.
HITRUST is the market’s answer to the question HIPAA does not fully answer: how do you prove to a counterparty credibly and without a bespoke audit that your security controls actually work?
For clinical and diagnostic labs specifically, the compliance stack is:
- HIPAA (mandatory)
- CLIA (mandatory)
- CAP (functionally mandatory for full-service labs)
- HITRUST (contractually mandatory for enterprise customers).
Onymos DocKnow is designed to reduce the documentation and data-handling burden across all of them at the layer where most compliance programs actually break: intake.
See what Onymos looks like in your lab’s workflow.
FAQs
Does HITRUST certification mean you’re HIPAA compliant?
Not automatically. HITRUST r2 certification maps extensively to HIPAA Security Rule requirements, and achieving r2 provides strong evidence of HIPAA compliance. However, organizations are still legally subject to HIPAA regardless of their HITRUST status, and a HITRUST certification that excluded certain systems or processes may not cover the full scope of HIPAA obligations.
Can a business associate use HITRUST certification to satisfy HIPAA BAA requirements?
Partially. HITRUST certification demonstrates that a vendor has independently validated security controls but a BAA is still legally required under HIPAA regardless of HITRUST status. Certification supplements the BAA; it does not replace it.
Is HITRUST only for healthcare organizations?
No. HITRUST originally targeted healthcare but expanded its scope to be industry-agnostic in 2019. Organizations in financial services, higher education, retail, and technology now pursue HITRUST certification, particularly when they want a single certification that maps to multiple regulatory frameworks simultaneously.